
MutaSight
Molecular Modeling applied to Precision Oncology
Discover how molecular modeling and bioinformatics tools are revolutionizing cancer treatment by enabling precise analysis of tumor mutations.


Discover how molecular modeling and bioinformatics tools are revolutionizing cancer treatment by enabling precise analysis of tumor mutations.

In-depth molecular analyses enable us to identify the mutations responsible for tumor development, so that we can target them more effectively.
Precision oncology aims to develop patient-specific treatments based on the genetic profile of each patient's tumor.
This personalized approach improves treatment efficacy and reduces side effects.
A mutation is a change in the DNA sequence of a gene. In some cases, it can lead to the development of cancer.
Depending on the type and location of the mutation, it may activate oncogenes or inactivate tumor suppressor genes.
Mutation analysis helps determine the best treatment options for each cancer patient.

By analyzing sequence differences between healthy and mutated proteins, researchers can detect abnormalities that contribute to cancer development.
By modeling the impact of identified mutations that are not documented in databases, we can assess how these changes affect the structure and function of proteins. These 3D analyses reveal how mutations may destabilize or alter the conformation of proteins, thereby disrupting their interactions and activity.
By identifying key proteins affected by mutations, oncologists can recommend targeted treatments that specifically inhibit their activity.
The case of a patient with uveal melanoma who responded to the treatment proposed by his oncologist, based on the results of Molecular Modeling of tumor mutations.
Next-Generation Sequencing (NGS) analysis of a biopsy from a patient with uveal melanoma detected several somatic mutations, some of which are not documented in databases.
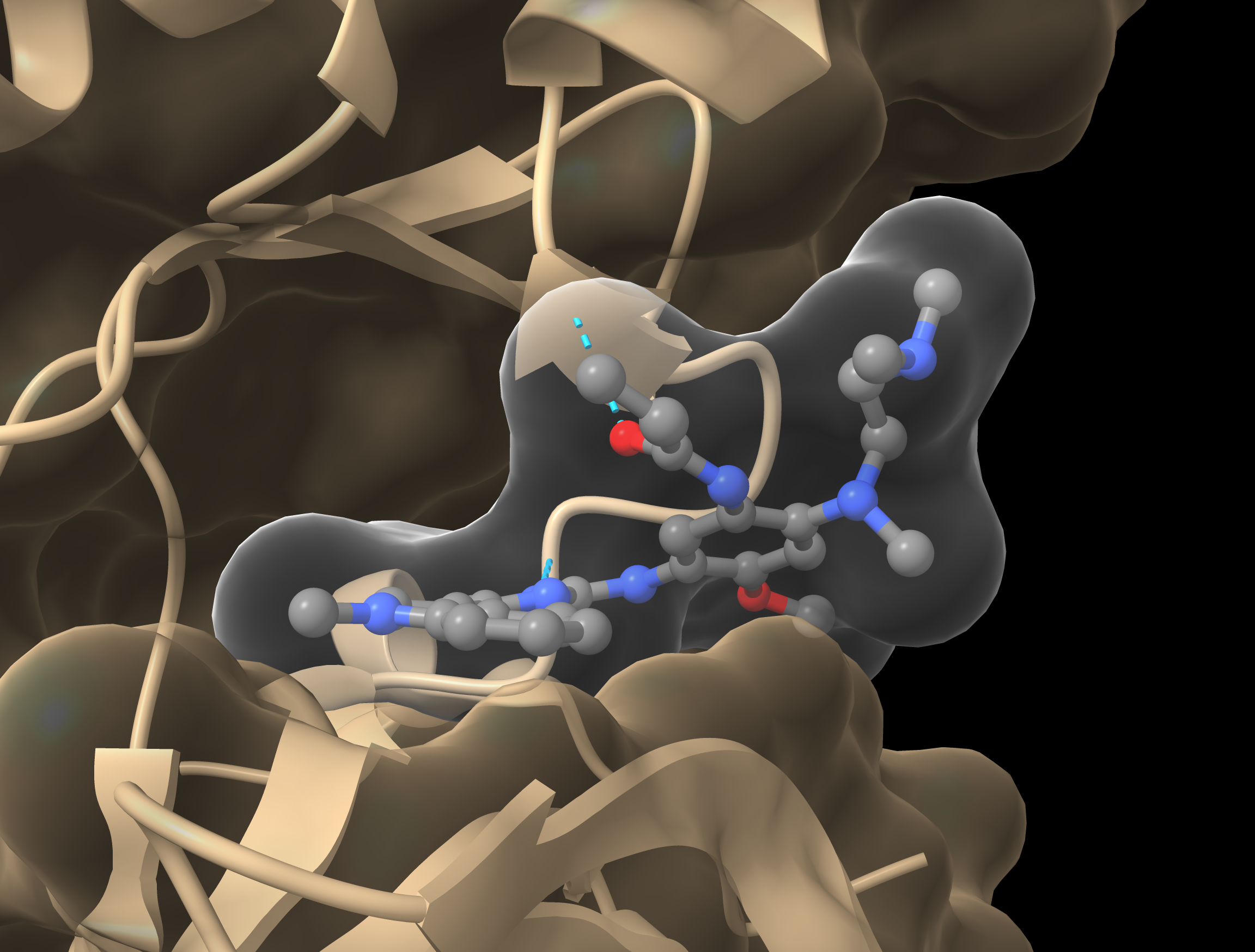
Molecular Modeling analysis of the unknown mutations predicted that two of them could be activating.
Inspired by the predictions from Molecular Modeling, the oncologist proposed a personalized treatment for the patient, who subsequently demonstrated a positive response to the therapy.
The details of this analysis have been published in a scientific journal available via this
link
Trametinib Induces the Stabilization of a Dual GNAQ p.Gly48Leu- and FGFR4
p.Cys172Gly-Mutated Uveal Melanoma. The Role of Molecular Modelling in Personalized Oncology
Fanny S. Krebs et al. Int. J. Mol. Sci. 2020, 21, 8021.
Abstract. We report a case of an uveal melanoma patient with GNAQ p.Gly48Leu who responded to MEK inhibition. At the time of the molecular analysis, the pathogenicity of the mutation was unknown. A tridimensional structural analysis showed that Gαq can adopt active and inactive conformations that lead to substantial changes, involving three important switch regions. Our molecular modelling study predicted that GNAQ p.Gly48Leu introduces new favorable interactions in its active conformation, whereas little or no impact is expected in its inactive form. This strongly suggests that GNAQ p.Gly48Leu is a possible tumor-activating driver mutation, consequently triggering the MEK pathway. In addition, we also found an FGFR4 p.Cys172Gly mutation, which was predicted by molecular modelling analysis to lead to a gain of function by impacting the Ig-like domain 2 folding, which is involved in FGF binding and increases the stability of the homodimer. Based on these analyses, the patient received the MEK inhibitor trametinib with a lasting clinical benefit. This work highlights the importance of molecular modelling for personalized oncology.